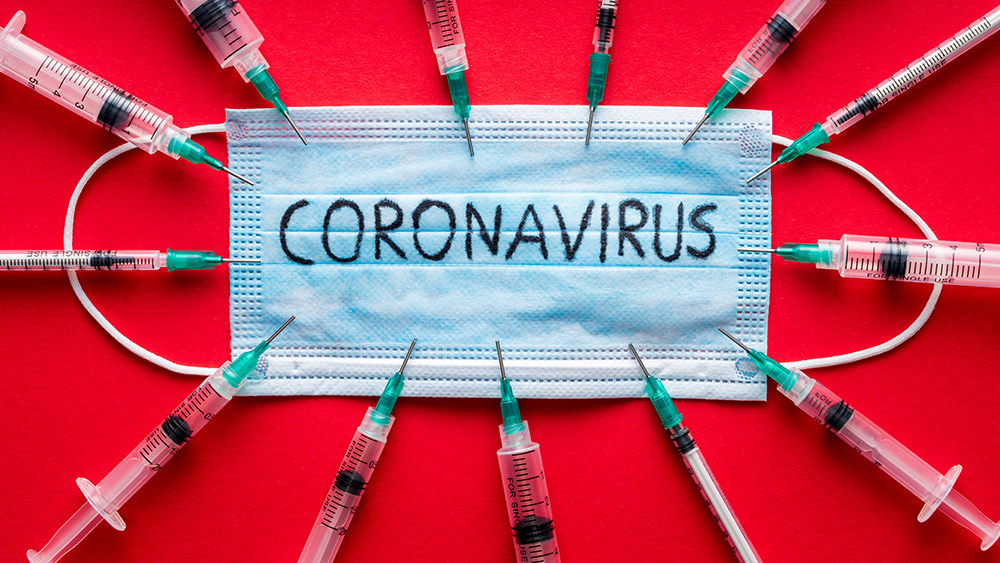
The Department of Defense’s (DOD) Joint Acquisition Task Force in partnership with the Department of Health and Human Services (HHS) has awarded ApiJect Systems America a lucrative $138 million contract to produce hundreds of millions of “smart” syringes for administering Wuhan coronavirus (COVID-19) vaccines to the masses.
According to the Pentagon, ApiJect subsidiary RAPID USA Inc. will immediately initiate Project Jumpstart, a program to develop a high-speed supply chain for prefilled syringes using blow fill-seal (BFS) plastics manufacturing technology. This will enable the “warp speed” development of hundreds of millions of medical-grade injection devices to be ready by October 2020.
“By immediately upgrading a sufficient number of existing domestic BFS facilities with installations of filling-line and technical improvements, ‘Jumpstart’ will enable the manufacture of more than 100 million prefilled syringes for distribution across the United States by year-end 2020,” says Pentagon spokesman Lt. Col. Mike Andrews.
By the end of 2021, ApiJect is planning to have the capacity to manufacture 330 million prefilled BFS syringes per month, meaning every person in American could theoretically be jabbed once per month with a new vaccine.
“RAPID USA is led by our multi-disciplinary team of experienced engineers, pharmaceutical technology experts, and management leadership,” says ApiJect Systems America CEO Jay Walker. “Our team is expending extraordinary efforts to ensure that when drugs are developed and tested all Americans can receive critical injections.”
“We will have done our part by providing the manufacturing capacity to support the necessary volume of ready-to-use prefilled syringes that contain essential medicines, be they therapeutics or vaccines.”
Coronavirus vaccine microchips will be able to send real-time data straight to the government
What makes these syringes “smart” is that each one comes with its own optional radiofrequency identification (RFID) chip or near-field communication (NFC) tag that can be synced with a smartphone app for tracking by healthcare workers. This will allow for the real-time transfer of data to the government about the syringe, what vaccine it contains, and who is receiving it.
“Before giving an injection, the healthcare worker will be able to launch a free mobile app and ‘tap’ the prefilled syringe on their phone, capturing the NFC tag’s unique serial number, GPS location and date/time,” the ApiJect website explains about the technology.
“The app then uploads the data to a government-selected cloud database. Aggregated injection data provides health administrators an evolving real-time ‘injection map.’”
While the technology is being unveiled in direct response to the Wuhan coronavirus (COVID-19) pandemic, ApiJect and RAPID say that it can also be used for all future pandemics or public health scares that drive the masses to demand rapid vaccination.
“RAPID will provide increased lifesaving capability against future national health emergencies that require population-scale vaccine administration on an urgent basis,” Andrews says.
At the onset, ApiJect will be manufacturing these “smart” syringes at manufacturing plants in Connecticut, Illinois, and South Carolina. As time goes on, this will expand to other locations throughout the U.S.
President Trump also recently announced the names of some of the folks who will be heading up “Operation Warp Speed,” the government initiative that will spearhead the delivery of the Wuhan coronavirus (COVID-19) vaccines to be inserted into ApiJect’s microchip-equipped syringes.
The top dog is former GlaxoSmithKline (GSK) executive Moncef Slaoui, who is also a general in the military. Another is Gustave Perna, another military general who will function as Operation Warp Speed’s chief operation officer (COO).
“I think we’re going to have a vaccine by the end of the year, and I think distribution will take place almost simultaneously because we’ve geared up the military,” Trump is quoted as saying about the timeline for the program.
Ethan Huff
Sources for this article include:
Related Posts
- Second AstraZeneca trial participant develops rare neurological condition following COVID-19 vaccination
- AstraZeneca’s COVID-19 vaccine trial halted following “serious adverse reaction” involving spinal inflammation
- Fast-tracked covid-19 vaccine alters human DNA, turns people into genetically modified property
- One in every 16 Irish boys has autism: Crisis worse than COVID-19 and nobody cares

