 The mechanism by which a cell dies, that is, apoptosis, necrosis, pyroptosis, or autophagic cell death, often depends on its ability to cope with the conditions to which it is exposed. The adaptive capacity of a cell ultimately determines its fate when it comes under stress. The strength or what we can call a cells adaptive capacity is directly related to nutritional sufficiency and proper cellular respiration that removes all toxins and wastes through the cell wall.
The mechanism by which a cell dies, that is, apoptosis, necrosis, pyroptosis, or autophagic cell death, often depends on its ability to cope with the conditions to which it is exposed. The adaptive capacity of a cell ultimately determines its fate when it comes under stress. The strength or what we can call a cells adaptive capacity is directly related to nutritional sufficiency and proper cellular respiration that removes all toxins and wastes through the cell wall.
Glutathione S-transferases (GSTs)2 are classified as a family of Phase II detoxification enzymes and are vital for cell function.
Cell wall permeability is in great part magnesium dependent[1] so when magnesium levels are low and calcium levels high cells walls become more rigid making it difficult for nutrients to enter and wastes to exit. Without magnesium, calcium cannot be properly absorbed and utilized, and muscles and cell walls tend to remain stiff. The secret of calcium-magnesium balance—as well as magnesium’s operation—is on a cellular level.
Low magnesium compromises the integrity of the cell wall. When magnesium is too low too much calcium and sodium floods into the cell causing hardening and overstimulation
Magnesium deficiency leads directly to cellular stress and that is why it is so important to include high dosages of magnesium chloride in every person’s protocol. Magnesium is vital to the cells; and just like in the emergency room, magnesium can mean the difference between life and death. Dr. Andrea Rosanoff, co-author of the book The Magnesium Factor with Dr. Mildred S. Seelig reported on a paper published back in 1926 by the British Journal of Experimental Biology where a scientist named C. F. A. Pantin was looking at amoeba cells. “When he placed some of the amoebas in a no-magnesium environment, they died. When he put them in a magnesium solution without calcium, they lived just fine but were unable to move. It was only when the amoebas were placed in both calcium and magnesium that they could actually move.”
The specific posttranslational modification of protein cysteine residues by the addition of the tripeptide glutathione is termed S-glutathionylation. This process is promoted by oxidative and nitrosative stress but also occurs in unstressed cells. – Dr. Danyelle M. Townsend
Cells can respond to stress in various ways ranging from the activation of survival pathways to the initiation of cell death that eventually eliminates damaged cells. Whether cells mount a protective or destructive stress response depends on many factors but the greatest would have to do with nutritional status. A healthy cell naturally chooses life but one that is already chronically stressed due to mineral and lipid deficiencies will have significantly less coping power, less resistance to stress. Other factors in calculating cellular resistance to stress are the nature and duration of the stress as well as the type of cells being affected.
Dr. Rosanoff points out that stress comes in many different forms. “There are emotional stresses, physical stresses and chemical stresses,” she explained. “Gravity is a stress. Any impact from the environment can be seen as a stress. There are internal stresses; for example, one cell inside your kidney could have a trigger coming from either a hormone or a nerve and that could be called, for that cell, a stress. A stress can be boiled down to something that makes a living thing react. When life is stressed, it needs to move, act, think, worry, tense. At the cellular level, the channels open and the calcium comes in, which fires off a series of reactions that allows the cell to respond to stress. We often call it the fight or flight reaction. If you have plenty of magnesium, you can go through these stressful events and go back down to your calm state and you’ll be in perfect health.”
We’re showing how the magnesium allows the calcium to rush into the cell, when required and that it’s the calcium inside the cell at critical levels that sets off activity. We then portray magnesium pushing the calcium back outside until it’s needed again. Every time they go through stress, the magnesium in the body is depleted more and more. – Dr. Andrea Rosanoff
The cell’s initial response to a stressful stimulus is geared towards helping the cell to defend against and recover from the insult so it behooves us to maximize the cells initial defensive response. That is why magnesium needs to be used before, during and after all surgeries because it is the key factor to how our total body physiology responds to the stress of surgery. Medical textbooks need to be rewritten because most doctors, medical professors and surgeons have not the slightest notion about putting these basic medical insights into clinical practice.
Magnesium and Cell Stress
The involvement of free radicals in tissue injury induced by magnesium deficiency[2] causes an accumulation of oxidative products in heart, liver, kidney, skeletal muscle tissues and in red blood cells.[3] Magnesium is a crucial factor in the natural self-cleansing and detoxification responses of the body. Magnesium protects cells from aluminum, mercury, lead, cadmium, beryllium and nickel, which explains why re-mineralization is so essential for heavy metal detoxification and chelation. Magnesium protects the cell against oxyradical damage and assists in the absorption and metabolism of B vitamins, vitamin C and E, which are anti-oxidants important in cell protection.
Glutathione requires magnesium for its synthesis.[4] Glutathione synthetase requires ?-glutamyl cysteine, glycine, ATP, and magnesium ions to form glutathione.[5] In magnesium deficiency, the ss y-glutamyl transpeptidase is lowered.[6] Data demonstrates a direct action of glutathione both in vivo and in vitro to enhance intracellular magnesium and a clinical linkage between cellular magnesium, GSH/GSSG ratios, and tissue glucose metabolism.[7]
Magnesium deficiency causes glutathione loss, which is not affordable because glutathione helps to defend the body against damage from cigarette smoking, exposure to radiation, cancer chemotherapy, and toxins such as alcohol and just about everything else. According to Dr. Russell Blaylock, low magnesium is associated with dramatic increases in free radical generation as well as glutathione depletion and this is vital since glutathione is one of the few antioxidant molecules known to neutralize mercury.[8]
SEE MORE about Magnesium Medicine
Life or Death?
If the stress goes beyond the cells’ adaptive capacity then the cells activate death signaling pathways. Therefore, depending on the level and mode of stress, different defense mechanisms and pro-survival strategies need to become the first concern of healthcare professionals. If ignored or worse, if cell conditions and stress are made worse by pharmaceutical’s toxicity (side effects), then the cell death programs are activated to eliminate these damaged cells from the organism. When this gets out of control the patient dies.
During tissue homeostasis there is equilibrium between the net growth rate and the net rate of cell death. A happy, healthy, well exercised nutritionally satisfied person will maintain equilibrium much easier whereas excessive negative emotions are a stress that puts physiological homeostasis in danger. When the cellular environment turns acidic and toxic we have generalized stress acting on all the cells and tissues in the body and then when an infection hits it can be the last straw that snaps the camel’s neck. The vast majority of the public have high levels of toxicity because of persistent exposure especially in cities and near highways; industrial parks, incinerators, and coal fired electric plants. We also gather toxicity from the water we drink and the food we eat.
Likewise when people are exposed to chemotherapeutic agents, radiation from medical tests and treatments, or other genotoxic agents such as heavy metals including mercury, polycyclic hydrocarbons, and a long list of hostile chemicals and drugs – damage to DNA is a common initial event. There are types of stress events that go for the heart of the cell, for the nucleus. Again we will find magnesium, selenium and bicarbonate very useful in buffering the cell to minimize damage to the DNA strands.
Avoiding such damaging agents and medical treatments and tests should be our first choice but unfortunately most licensed physicians are not given a choice of treatment when it comes to diseases like cancer. Treatments and even diagnostic procedures are stressful on cells and it is no wonder why modern oncology hurts so much. Modern medicine is guilty of overusing both tests and treatments that cause cancer in their attempt to cure it, which does not make a lot of sense, especially to our cells that bear the brunt of the toxicity we are being exposed to.
MORE ABOUT Natural Chelation
The key question is what is involved with the switch from prolife defensive responses to cellular suicide? Is it a molecular switch fixed on a certain point or is it on some moving line? Is there a formula or medical model to completely understand this? Unfortunately no, this is new to medical science but it’s crystal clear that magnesium is at the heart of it as is selenium because of their direct relationship with glutathione levels.
If we are going to talk about models it’s going to have to be quite encompassing to even include something as basic and common as dehydration (H2O deficiency). Water certainly needs to be hydrated into our cellular stress models. We know already that much damage can be prevented with high levels of glutathione. The trick is in knowing how best to raise these levels.
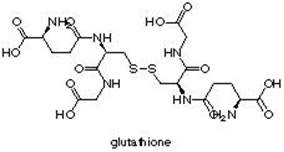
Gutathione (gl?’t?-th?‘?n’) is a polypeptide, C10H17N3O6S, of glycine, cysteine, and glutamic acid.
Spirulina is effective in raising glutathione levels because it supplies glutamic acid, glycine, cystine, and methionine in easily absorbable forms. Cysteine and cystine are closely related. One cystine molecule is composed of two bonded cysteine molecules and each can convert to the other as required. Both amino acids contain sulfur (via free sulfhydryl groups) which makes them antioxidants. See my chapter on Superfoods for Cellular Survival.
Glutathione concentrations are sensitive to diet and nutritional status.Strategies to rapidly restore glutathione for both antioxidant and immune defense systems are crucial especially in malnourished patients. Spirulina and other live green superfoods, magnesium, selenium, Vitamin C, s-adenosylmethionine (SAMe), N acetyl cysteine (NAC) and Alpha lipoic acid (ALA) are all concentrated nutritional medicines that will raise glutathione levels. “For every molecule of pesticide that your body’ detoxifies, you throw away or use up forever, a molecule of glutathione, magnesium and more,” says Dr. Sherry Rogers who goes on to say that, “Your body uses nutrients to make this glutathione and it uses up energy as well. Every time we detoxify a chemical, we use up, lose, throw away forever, a certain amount of nutrients.”
The James study shows that children with regressive autism have consistently elevated levels of oxidative stress as compared to normal healthy children. Individuals with reduced glutathione antioxidant capacity (magnesium and selenium deficient) will be under chronic oxidative stress and will be more vulnerable to toxic compounds that act primarily through oxidative damage. Glutathione has many profound roles in the body. One role is to enable the liver to remove toxins, medications and other substances from the body. Without it, these substances cannot be removed properly so they accumulate in the blood stream and in all the tissues of the body.
Selenium
When mercury “binds” to selenium or cysteine it is no longer free to “bind” to anything else — like brain or kidney tissue. The detoxifying effect of selenium on mercury toxicity is due to a formation of a biologically inactive complex containing the elements in an equimolar ratio. The complex is unable to pass biological barriers, placenta and choroid plexus and is stored in the liver and the spleen, even in the brain in a non toxic form. Mercury is a direct threat to cells causing stress of prolonged duration. Since mercury toxicity is a principle threat coming into to our biological sphere via air, water, food, dental fillings and vaccination shots it’s important to know and use selenium to reduce cellular stress.
Selenium is essential for our immune system to function at optimal performance as it has a direct relationship to cells adaptive cellular capacity. Importantly selenium has been directly correlated with cancer rates. Dr. Shu-Yu Yu measured the selenium content of blood stored in blood banks in 30 different regions in China, and classified the regions as high selenium, medium selenium, and low selenium. They then compared death rates from cancer to the selenium rates and found there was an exact correlation. In the low selenium classification, three times as many people died from cancer as in the high selenium classification.
Dr. Richard Donaldson of the St. Louis Veterans’ Administration Hospital conducted a clinical trial with terminally ill cancer patients. He found that when he could raise the patients’ blood levels of selenium into the normal range, their pain and tumor sizes were often reduced. In a 140 patient study of cancer victims treated with selenium, Dr. Donaldson reported in 1983 that some patients deemed terminal with only weeks to live were completely free of all signs of cancer after four years; all the patients showed a reduction in tumor size and in pain.
Dr. Harold Foster has stated that death rates in the USA for breast, colon, rectal and lung cancer are lower when blood selenium levels are high. Dr. Foster reported that cancer patients with low selenium levels tend to have a wider spread of the disease, more recurrences and die sooner.[9]
Protein S-glutathionylation
Different environments may promote different S-glutathionylation patterns.
Dr. Danyelle M. Townsend of the Department of Pharmaceutical and Biomedical Sciences, Medical University of South Carolina tells us that protein S-glutathionylation is the reversible formation of a mixed-disulfide between glutathione and protein thiols. Medical scientists are breaking into new areas of molecular biology and don’t quite understand everything they are discovering so don’t feel inadequate if you cannot quite wrap your mind around protein S-glutathionylation.
SEE MORE about GLUTATHIONE MEDICINE
S-glutathionylation is involved in protection of protein cysteines from irreversible oxidation. It is involved in protein redox regulation, storage of glutathione, and protection of protein thiols from irreversible oxidation. In one study[10] it was shown that glutathione (GSH) depletion, reactive oxygen species generation, hyperpolarization of mitochondrial transmembrane potential (DeltaPsim) and formation of protein-GSH mixed disulphides (S-glutathionylation) are early molecular events that precede the induction of cell death.
Other studies have implicated S-glutathionylation as a cellular response to oxidative/nitrosative stress thus altered levels of S-glutathionylation in some proteins have been associated with numerous pathologies, many of which have been linked to redox stress in the endoplasmic reticulum (ER). Conditions within the ER affect rates of S-glutathionylation.
S-glutathionylation increases; during cataractogenesis, some of lens proteins, including ?- and ?-crystallins, form both mixed disulfides and disulfide-cross- linked aggregates, which increase with cataract severity.
While conditions of stress stimulate cells to mount protective responses to counteract the effect of the stress on cellular processes, if the stress remains unresolved, eventual death of the cell ensues. This raises key questions about the molecular mechanisms involved in this switch from prosurvival signaling to prodeath signaling. The point is that glutathione levels need to be maintained at the most optimum levels if we want to keep on track for surviving the toxicity of the 21st century.
Concluding
Remaining in the “keyed-up” calcium filled condition is unhealthy for the cells. Under ideal circumstances, once the stress has ceased, magnesium, if there is enough of it, helps to push the calcium back outside the cell, allowing the cell to “calm down.” As a result the nerves stop firing, muscles cease contracting and adrenaline is no longer secreted and we relax. Magnesium was correctly called the beautiful medicine by the ancient Chinese for from a molecular biology point of view the metal is priceless. For oncologists it is important to know that magnesium also has a hand in protecting the DNA and is a key ion in cell division.
Likewise pH control is a key to cellular survival and determines much of a cells adaptive capacity relative to stress. Iodine, selenium and even zinc are not far behind magnesium in relationship to increasing cell strength. Eating as low on the food chain as possible is also a key with spirulina or spirulina based products essential for increasing adaptive cellular capacity.
Dr. Mark Sircus
References (10)
- That magnesium ions can control the porosity and stability of cell membranes has been suggested by several investigators. Mg2+ is known to stabilize bacterial cytoplasmic membranes and to cause a significant reduction in the porosity of the cytoplasmic membrane of Bacillus megaterium.
- Magnesium deficiency (MgD) has been associated with production of reactive oxygen species, cytokines, and eicosanoids, as well as vascular compromise in vivo. Although MgD-induced inflammatory change occurs during “chronic” MgD in vivo, acute MgD may also affect the vasculature and consequently, predispose endothelial cells (EC) to perturbations associated with chronic MgD. As oxyradical production is a significant component of chronic MgD, we examined the effect of acute MgD on EC oxidant production in vitro. In addition we determined EC; pH, mitochondrial function, lysosomal integrity and general cellular antioxidant capacity. Decreasing Mg2+ (< or = 250microM) significantly increased EC oxidant production relative to control Mg2+ (1000microM). MgD-induced oxidant production, occurring within 30min, was attenuated by EC treatment with oxyradical scavengers and inhibitors of eicosanoid biosynthesis. Coincident with increased oxidant production were reductions in intracellular glutathione (GSH) and corresponding EC alkalinization. These data suggest that acute MgD is sufficient for induction of EC oxidant production, the extent of which may determine, at least in part, the extent of EC dysfunction/injury associated with chronic MgD. Effect of acute magnesium deficiency (MgD) on aortic endothelial cell (EC) oxidant production.Wiles ME, Wagner TL, Weglicki WB. The George Washington University Medical Center, Division of Experimental Medicine, Washington, D.C., USA. mwiles@nexstar.com Life Sci. 1997;60(3):221-36.
- Martin, Hélène. Richert, Lysiane. Berthelot, Alain Magnesium Deficiency Induces Apoptosis in Primary Cultures of Rat Hepatocytes.* Laboratoire de Physiologie, et Laboratoire de Biologie Cellulaire, UFR des Sciences Médicales et Pharmaceutiques, Besançon, France. 2003 The American Society for Nutritional Sciences J. Nutr. 133:2505-2511, August 2003
- Linus Pauling Institute lpi.oregonstate.edu/infocenter/minerals
- Virginia Minnich, M. B. Smith, M. J. Brauner, and Philip W. Majerus. Glutathione biosynthesis in human erythrocytes. Department of Internal Medicine, Washington University School of Medicine, J Clin Invest. 1971 March; 50(3): 507–513. Abstract: The two enzymes required for de novo glutathione synthesis, glutamyl cysteine synthetase and glutathione synthetase, have been demonstrated in hemolysates of human erythrocytes. Glutamyl cysteine synthetase requires glutamic acid, cysteine, adenosine triphosphate (ATP), and magnesium ions to form ?-glutamyl cysteine. The activity of this enzyme in hemolysates from 25 normal subjects was 0.43±0.04 ?mole glutamyl cysteine formed per g hemoglobin per min. Glutathione synthetase requires ?-glutamyl cysteine, glycine, ATP, and magnesium ions to form glutathione. The activity of this enzyme in hemolysates from 25 normal subjects was 0.19±0.03 ?mole glutathione formed per g hemoglobin per min. Glutathione synthetase also catalyzes an exchange reaction between glycine and glutathione, but this reaction is not significant under the conditions used for assay of hemolysates. The capacity for erythrocytes to synthesize glutathione exceeds the rate of glutathione turnover by 150-fold, indicating that there is considerable reserve capacity for glutathione synthesis. A patient with erythrocyte glutathione synthetase deficiency has been described. The inability of patients’ extracts to synthesize glutathione is corrected by the addition of pure glutathione synthetase, indicating that there is no inhibitor in the patients’ erythrocytes.
- Braverman, E.R. (with Pfeiffer, C.C.)(1987). The healing nutrients within: Facts, findings and new research on amino acids. New Canaan: Keats Publishing
- Barbagallo, M. et al. Effects of glutathione on red blood cell intracellular magnesium: relation to glucose metabolism. Hypertension. 1999 Jul;34(1):76-82. Institute of Internal Medicine and Geriatrics, University of Palermo, Italy. mabar@unipa.it
- http://www.dorway.org/blayautism.txt
- Foster HD. “Landscapes of Longevity: The Calcium-Selenium-Mercury Connection in Cancer and Heart Disease,” Medical Hypothesis, Vol. 48, pp 361-366, 1997.
- Biochim Biophys Acta (2006) 1763: 214-25. GSH depletion, protein S-glutathionylation and mitochondrial transmembrane potential hyper. A Di Stefano, S Frosali, A Leonini, A Ettorre, R Priora, FC Di Simplicio, P Di Simplicio
 Schwerdtle, Cornelia, and Arnoul, Franz. Introduction into darkfield diagnostics.
Schwerdtle, Cornelia, and Arnoul, Franz. Introduction into darkfield diagnostics.
OMAX 40X-2500X 1080p HDMI Camera Darkfield Lab Siedentopf Trinocular LED Microscope for Live Blood


